The connection of Silicon Valley to neurotechnology includes: projects inside name-brand technology companies, new companies with audacious goals, and the movement of talent and funding.
In the first part of “What Silicon Valley needs to know about the brain, and what it doesn't” I focused on the question: How could Silicon Valley culture transform technologies already worked on (by academics and medical device companies) for decades? Here I take a step back to consider some definitions of key terms: electrodes, minimally-invasive, non-invasive, and safe.
Definitions don't make or break technology (except in special cases of government regulations). And definitions can be application-specific, varied, and changing. The scientific philosopher Karl Popper thought very little of definitions: “People who talk too much about words…or definitions, they do not actually put anything forward…and they certainly don't help with clarity.” The physicist David Deutsch, who followed in Popper tradition on this, called instead for clear explanations.
When certain key words are thrown around with special importance in neurotechnology, it’s good to consider what that meaning is. My organization around a few definitions (electrodes, minimally-invasive, non-invasive, and safe), is about explaining further how neurotechnologies work.
Electrode
The word electrode comes up a lot in neurotechnology. Better electrodes are part of the sales pitch of almost any brain-computer-interface (BCI) venture, such as more, smaller, more flexible, denser-packed electrodes. In the Neuralink Fall 2022 “show and tell”, DJ Seo, VP of Brain Interfaces (following up Elon Musk’s opening remarks) explains “…this is a tractable engineering challenge since everything about your intentions, your thoughts, your experiences are in your brain encoded as binary statistics of action potentials. If you’re able to put electrodes in the right places with the right sensing and stimulation capabilities…”
What is an electrode? The electrodes are any (and the only) places on a neurotechnology device where information, carried by electricity, is transferred between the device and the body. When the electricity flows in the direction from the body to the device, information from the brain is “read” by the device. When electricity flows in the direction from the device into the body, this is called electrical stimulation - and we “write” information unto the brain.
Individual electrodes represent, in theory, independent locations for read/write. So the more electrodes you have, the more channels of communication between the device and brain. Very small electrodes (on the scale of a mico-meters) can in theory interface with the individual working units of the brain: neurons. In this case, the ongoing electrical activity of each neuron, called action potentials, can be detected as brief spikes. The timing of these spikes (plotted as vertical dashes) forms a read-out of each neuron’s activity, like this:

The image above shows the independent action potential (spikes) of three neurons over time, as detected by three electrodes. Each electrode detects the activity of only the neuron it is close to (assuming no cross-talk). In principle each thought you have (including intention or memories) is represented in the brain by an action potential pattern in a given set of neurons. Therefore, thoughts can be read by measuring their underlying neuronal activity – provided this neuronal code can be decoded into thoughts (e.g., intentions).
Larger electrodes (mm to cm) interact with a large group of neurons from a volume of the brain. Larger electrodes can be farther away from the neurons they sense, and the activity recorded from each larger electrode is therefore an average of activity from many neurons that it senses. Larger electrodes are considered most stable over long periods of time (a decade or more), though not without risk of complications. The information collected from large electrodes (neuron groups) is not necessarily less useful than information from micro-electrodes (single neurons); this depends entirely on the device application and the level of explanation desired from the decoding. Similarly, all existing successful therapies using electrical stimulation use relatively large electrodes. And the value of increasing stimulation electrode density may belimited by increasing complexity in using them optimally.
The electricity in the body is carried by moving salts dissolved in the brain soup (like sodium and chloride ions that result from pouring table salt into water) while in electronics (computers, wires, etc.) electricity is carried by moving electrons. The electrode is therefore where a transformation occurs from ion-carried (body side) to electron-carried (device side) electricity. For reading brain activity this transformation is facilitated by using special amplifiers. For writing brain activity, careful attention is needed so that this transformation does not produce unwanted electrochemical products. A critical safety concern for electrical stimulation with decreasing electrode size or new electrode materials.
Among the hardest technical issues comes from the body being a rather inhospitable environment for electrodes, for example because of ever-present brain movement (including blood flow pulsation, breathing, etc.) or the cruding up of the electrode surface. These problems can get harder with ever smaller electrodes.
If you want to find the electrodes on a neurotechnology device look for anywhere there is exposed metals exposed to the body. For non-invasive devices there may also be a salty gel (like an adhesive hydrogel electrode) between the metal and the skin. Electrodes are often clustered together on the ‘business end’ of the device that is closest to the body part we want to interface with. The electronics used for recording, stimulation, data processing and transmission are connected to, but are separate components (as some distance) from the electrodes.
For neurotechnologies that don't rely on electricity for interfacing, electrodes are replaced with different types of sensors and transducers. Such as those using light or ultrasound.
Minimally Invasive (Neurotechnology)
Brain interface technology can be non-invasive or invasive, only the latter involves implanting electrodes inside the body / brain. The reason to get the electrodes closer to the brain, is to be able to better resolve (read/write) the activity of the brain. Think of using a set of microphones (electrodes) to listen to a dense crowd (brain full of neurons) – it’s more useful to get individual microphones close and ideally inside the group in order to hear distinct groups of speakers (neurons).
Companies like Precision, Neuralink, Blackrock Neurotech, and Paradromics will implant the electrodes by drilling burr holes in the skull. The electrodes must be connected to a computer processor with battery power, which can either be fully implanted (and wirelessly connected to an external device) or connected using an exposed plug. Neuralink fully implants their transmitter (the “link”) in a cavity drilled out of the skull. Paradromics appears to be taking a similar approach with a “cranial hub” that “sits in-line with the skull” with a further “transceiver” implanted near the chest. The BlackRock Neurotech systems connect through a plug exposed on the scalp.
To increase access to their technologies, these companies share a vision where reducing invasiveness goes along with reduced cost and hospital burden. Craig Mel, Precision’s President and Chief Product Officer hopes for “essentially a sub-mm thick” burr hole that is eventually “compatible with an outpatient procedure.” Consistent with Precision’s central focus on minimally invasive. Like Neuralink and other companies, Precision hitches their innovation not simply on the device itself, but the associated novel surgical procedure to implant it.
Contrasting with existing brain implants is naturally a selling point for all these companies. In “What makes us different” Neuralink says “For example DBS leads have only 4-8 electrodes and are about 800 times larger in diameter.” DBS stands for Deep Brain Stimulation, an established FDA cleared treatment provided by medical device stalwarts like Medtronic, Abbott, and Boston Scientific. DBS burr holes are about 1.5 cm or less, which is a little larger than the diameter of the implanted electrode and also allows for a lead anchoring device. In contrast, the Neuralink ‘link’ is about 3 cm, though the component carrying the electrodes is smaller. The DBS electrodes are connected by a wire, tunneled under the scalp, to a device implanted under the chest (the “can”) which houses the battery, electronics, and wireless communication to a remote and/or charger.
The electrodes, and the small wires that connect them to the device electronics, are mechanically supported by a non-conductive material. The electrodes, the wires, and this support material can be referred to as the lead. A group of electrodes on a lead may be called an electrode array. Precision is basing their platform on a lead shaped as a thin sheet that is placed on the cortical surface, the “Layer7 Cortical Interface”. Electrodes arranged along the brain surface are generally called Electrocorticography (ECoG). Neuralink has electrodes on “Neural Threads” that are woven into the brain. Paradromics rely on small three-dimensional arrays of electrodes, each row of electrodes arranged on a needle, and the needles pressed about a cm into the brain surface. BlackRock Neurotech works with needle array electrodes (Utah Array), cortical surface electrodes, and deep penetrating electrodes. Deep electrodes, like those used in DBS, rely on (partial) ring electrodes arranged along long penetrating leads.
Neuropace provides an FDA cleared system to both record and stimulate the brains of patients with epilepsy (“RNS”) using a combination of cortical surface (ECoG) and DBS leads. The device with electronics is implanted “flush with the skull”. Intermittently data from the devices can be collected wirelessly through the scalp. The expression minimally invasive does not feature on the Neuropace website, though the system is marketed as “less invasive than resection surgery.” Medtronic also provides devices for both recording and deep brain stimulation.
Sychron, a clinical trials stage company, takes a different approach as an “endovascular brain computer interface”. Their electrodes are implanted on a “stentrode” which like a stent is implanted not through the skull but guided through blood vessels. As a result, the electrode ends up not touching neurons, but adjacent on the inside of brain blood vessels. Technology uniqueness and challenges aside, here again the implant procedure is central to achieve minimal-invasiveness.
Dr. Parag Patil, a biomedical engineer and neurosurgeon at University of Michigan, shared his perspective with me:
When thinking about “invasiveness,” one naturally focuses on the size of a hole in the skull or incision in the skin. But when the goal is to minimize invasiveness, we really mean minimizing long-term risk, and a focus on size may not be helpful. For example, a longer and gently curved incision, along skin’s lines of Langer, will heal better than a smaller more curved incision. What matters more to me is reducing risks related to that brain region that will be interfaced with – considering how its function might be impacted as well as the function of any impacted blood vessels.
The term “minimally invasive” is always relative to something else. At some level any incision is invasive and the least invasive strategy always to avoid surgery! Some procedures, like ablation of brain tissue using ultrasound, are enormously “invasive” but have no incision or burr hole at all.
Now, whether the same surgery is described as “minimal” or “moderate” or “very” invasive does not change the actual procedure or its associated risk. At the same time, it does hugely, and understandably, impact the patient’s perspective and anxiety towards the surgery. My role as a neurosurgeon is to educate, not to convince patients. So, I do my best to give the patient a full and accurate understanding of the process.
Dr. Patil further suggested that it is not useful to focus on or pigeon-hole a definition of minimally invasive. Rather, physicians plan and explain procedures to maximize patient outcomes. This echo’s Karl Popper’s depreciation of reliance on definitions.
Instead of interfacing with the brain, neurotechnologies can interface with the spinal cord or nerves –parts of the nervous system outside the skull. One form factor is electrodes implanted next to the target in the spinal cord or nerves, connected by a cable to a battery-powered device (the “can”) implanted in a more convenient body location. Spinal Cord Stimulation (SCS) is FDA cleared for pain and under investigation for rehabilitation following injury. Saluda Medical provides a SCS system that both stimulates and records (reads) from the spinal cord.
Peripheral Nerve Stimulation (PNS) is FDA cleared for pain and under investigation for a range of disorders including bladder dysfunction, inflammatory disease, hypertension, diabetes, and arthritis. Galvani Bioelectronics (formed through a partnership between GlaxoSmithKline (GSK) and Verily Life Sciences (formerly Google Life Sciences) has developed a peripheral nerves stimulation platform that is “designed to be inserted via a minimally invasive laparoscopic procedure with the intent to reduce patient discomfort and recovery times.” Established companies in SCS and PNS usually do not tout ‘minimally invasive’ as a distinguishing feature of their products (instead focusing on special energy delivery and mechanisms such as low-frequency by Presidio Medical or high-frequency by Nevro), as SCS and PNS procedures can already be out-patient.
Another approach is to implant a device with limited electronics or battery, and then control and power the device using a wearable. Nalu Medical provides a “minimally invasive” SCS and PNS device with the “smallest footprint” compared to other commercial implanted stimulators, by using a remote-control wearable worn over the implant location. Inner Cosmos is promising a “digital pill” the size of “two stacked pennies” implanted under the scalp intended to treat depression by stimulation through the skull, when in temporary use the device is powered by a second device held over the skin. Stimwave technology (sold in 2022 following bankruptcy) minimized its implanted PNS technology with control and power coming from a “Wearable Antenna Assembly (WAA)”. Almost all brain-reading (BCI) companies externalize their decoding software, except in cases where closed-loop stimulation requires fast on-board processing (as the case for Neuropace and Saluda Medical).
Some techniques insert a percutaneous electrode for a limited time under the skin next to the nerve. This includes electroacupuncture or subdermal needle electrodes for recording specific nerves or muscles. The electrodes are connected to an external recording or stimulation device.
In summary, in the context of neurotechnology minimally invasive typically implies an implanted device where the surgical procedure is intended to minimize potential complications, damage to tissue (e.g., skull burr hole size), and/or hospital stay. But minimally invasive is used in a relative context, and any definition does not substitute explicating technology and procedures.
Non-Invasive (Neurotechnology)
A non-invasive neurotechnology device is one whose use does not involve breaking the skin or physically entering the body, including a body opening. This definition can apply equally to the device itself or methods of use (absence of surgical procedures). Examples of non-invasive neurotechnologies would be caps with electrodes held to the scalp which can be used for recording electrical signals from the brain (EEG) or stimulation the brain externally with electricity (transcranial electrical stimulation). Non-invasive brain interfaces can also use light for recording (fNIRS) and stimulation (transcranial photobiomodulation). Ultrasound can also be used for imaging and stimulation.
Non-invasive interfaces can be applied anywhere on the surface of the body. Commonly Transcutaneous Electrical Nerve Stimulation (TENS) is used to manage pain and physical rehabilitation. Stimulation on the external ear is non-invasive. In approving the PoNS device using tongue stimulation, the FDA adopts the expression "non-implantable device". Inserting an electrode inside the mouth would fall outside this non-invasive definition.
Despite this pretty straightforward definition there have been alternative proposals, specifically in the context of neurotechnology, to adopt more complex and ambiguous definitions on non-invasive. MIT Technology Review describes that anything “eliciting some sort of change in the brain”, or “changes the way a person’s brain works”, or has any side-effects, or (following a public survey) “involve multiple trips to the doctor’s office are invasive because they impinge on a person’s time”. Such definitions, by allowing everything, exclude nothing- and so I don't think useful.
The concept that the body responds to non-invasive electric stimulation dates back to the discovery of electricity itself. Since the earliest human experience of electricity came from feeling electric shocks. The word electricity comes from Greek electron which means amber. Rubbing amber can generate an electrostatic charge that can be felt or attract light objects like feathers. It seems the first use of electric and electricity in the English language followed this notion of an ability to attract.
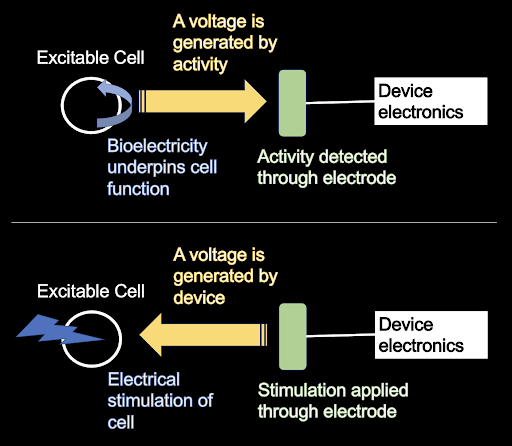
Luigi Galvani (1737-1788) discovered that rudimentary batteries could animate (produce movement) in animals, or even just isolated parts of animals like a frog’s legs. This encouraged the study of what we now understand as bioelectricity – how the body uses electricity as an internal signal. It is no coincidence that those cells that use of bioelectricity (such as action potentials) are the most sensitive applied electrical stimulation. Excitable cells include neurons, cardiac cells, and skeletal muscle cells- these cells depend on electrical activity for their functions and – for precisely this reason – are easily excitable by electrical stimulation (An older expression for such cells was irritable cells). This reciprocity is show in the figure below:
The development of early batteries by Alessandro Volta (1745-1927) - from which we get the modern unit of Volts- likely involved testing how much of a shock he could produce on himself and others. The more volts (more stacks in the voltaic pile), the bigger the shock. Both Galvani and Volta became interested in the therapeutic uses of electricity. Work with voltaic piles was quickly investigated by others to produce involuntary muscle twitches and noninvasive brain stimulation in patients. The idea of medical electricity is much older still. Including the use of electric eels by the ancient Egyptians and the use of electrostatic generators by Benjamin Franklin. The devices used today (which include computers to precisely regulate where and how electricity is applied through specially designed electrodes) are as distinct from these old stimulators as modern pharmacotherapy is from early plant-based medicine. In my view, the ability to formulate targeted electrical stimulation recipes to heal the body is as rational and unlimited in potential as chemical-based therapies. But I digress.
The discovery that brain electricity can be recorded non-invasively from the scalp dates to the 1920’s by a German psychiatrist named Hans Berger. As a young man, Berger suffered an accident with a horse. His sister, at home many miles away, had a feeling he was in danger that was so strong she insisted their father telegram him. The incident made such an impression on Berger that, years later in 1940, he wrote: "It was a case of spontaneous telepathy in which at a time of mortal danger, and as I contemplated certain death, I transmitted my thoughts, while my sister, who was particularly close to me, acted as the receiver." Berger first used electrodes made of wire inserted under the scalp (minimally invasive) and switched to non-invasive electrodes attached to the scalp. The technique we now call electroencephalography (EEG). Berger observed several spontaneous electrical oscillations generated by the brain, the most noticeable among them we now call alpha waves. Alpha waves broadly indicate a person’s level of vigilance vs relaxation, mistake prediction, and certain types of memory recall. Modern EEG measures many types of brain oscillations as well as other forms of brain signals.
How can an electrode placed on the scalp record activity from neurons, on the other side of the skull, in the brain? Large electrodes (in general but especially when at a distance from the brain) rely on a principle of summation. One neuron on its own does not generate a strong enough signal to be detected by a large electrode at a distance. But when many neurons generate activity together, the ‘voices add together’, similar to a bunch of people singing together so they can be heard across a large concert hall. So large electrodes at a distance will only pick off signals from many neurons active in synchrony. This is shown in the cartoon below:

There is a reciprocity between the resolution of an electrode as far as its recording (from a single neuron to an entire brain region) and its resolution of stimulation. So, electrodes that are smaller and closer to neurons can both record and stimulation with more selectivity. Non-invasive recording technology overcomes this limitation by using many electrodes across the scalp that can re-construct what is happening inside different brain regions (this is called source localization). Modern EEG systems used many electrodes (dozens or even hundreds) to both allow recording from across the whole brain and to resolve activity from individual brain regions. Advances in non-invasive electrical stimulation (including High-Definition transcranial Electrical Stimulation developed in my lab) optimize current steering to targeted brain regions. But for both recording and stimulation breakthroughs in usability (such as reading thoughts or treating a brain disease) derive not simply from increasing resolution, but from understanding how brain function impacts the use case. An example includes work from Dr. Robert Reinhart who artfully combines EEG and High-Definition stimulation to enhance memory.
Aside from electricity, other forms of energy can be passed through the scalp and skull for non-invasive brain recording (read) or stimulation (write). This includes ultrasound and light. Kernel was founded in 2017 by Bryan Johnson, formally CEO of Braintree (which acquired Venmo and was in turn acquired by PayPal). Kernel is developing a non-invasive brain recording device using Time-Domain functional Near-Infrared Spectroscopy (TD-fNIRS). The near-infrared means light. Openwater was founded in 2016 by Mary Lou Jepsen, formally at Facebook and Google X. Open-water is developing a non-invasive brain recording (read) and stimulation (write) device based on physics of light. In her talks, Jepsen makes the point by shining a laser pointer through her finger, a beaker a blood, or a fresh cut of meat. The use of light to monitor the brain has been studied for decades using functional Near-Infrared Spectroscopy (fNIRS). The use of light to stimulate the brain is also decades old, once called Low Level Light Therapy (LLLT) but now referred to as transcranial photobiomodulation (tPBM). How can light active the brain? Mitochondria (a part of cells that produce the energy molecule ATP) have been proposed as likely sites for the initial effects of therapeutic light, leading to increased cell energy and then activation of brain growth factors. My colleague Jacek Dmochowski has suggested that tPBM enhances connectivity between brain regions.
In addition to stimulation through the skull some technologies activate cranial nerves. Cranial nerves are extensions of the brain that cross outside the skull. Most nerves in the body, especially periphery, are peripheral nerves, which junction into the spinal cord. Non-invasive technologies can be used across the entire body. Cala Health provides a wrist worn treatment device for tremor relief, which provides electrical stimulation to the nerve running down the wrist.
In summary, in this context of neurotechnology, non-invasive is commonly understood as a device whose use avoids breaking the skin or putting an object inside a cavity. Function is therefore achieved by transmitting or receiving energy through the skin to a part of the nervous system. The effects on the body / brain may be nuanced or profound. A device with any implanted component would not be non-invasive.
Safe (Neurotechnology)
Everything has risks. In medical devices, the expression non-significant-risk (NSR) can be understood as the risk level associated with daily activities. Like driving, making toast, or playing ping-pong.
The FDA defines non-significant-risk by what it’s not:
A. What is a Significant Risk Device Study?
• Is intended as an implant and presents a potential for serious risk to the health, safety, or welfare of a subject; • Is purported or represented to be for use supporting or sustaining human life and presents a potential for serious risk to the health, safety, or welfare of a subject; • Is for a use of substantial importance in diagnosing, curing, mitigating, or treating disease, or otherwise preventing impairment of human health and presents a potential for serious risk to the health, safety, or welfare of a subject; or • Otherwise presents a potential for serious risk to the health, safety, or welfare of a subject.
B. What is a Nonsignificant Risk Device Study?
A Non-Significant Risk device study is one that does not meet the definition for a Significant Risk device study.
Note this definition is for a device study, not for the device itself. This distinction is important. The same device may be a significant risk in one use-case, and not in another. Any trial with an implanted device (including minimally-invasive) is significant risk by this definition.
In a guidance for low risk general wellness devices, the FDA indicates:
If the answer to any of the following questions is YES, the product is not low risk
1) Is the product invasive? (For purposes of this guidance, “invasive” means penetrates or pierces the skin or mucous membranes of the body.) 2) Is the product implanted? 3) Does the product involve an intervention or technology that may pose a risk to the safety of users and other persons if specific regulatory controls are not applied.
In the last case the FDA is indicating that a device may be low risk provided ‘regulatory controls’ (processes and tests the product undergoes), but not otherwise. Understanding FDA regulations can feel like reading tea leaves – which reflects the FDA allowing itself flexibility of enforcement. The FDA’s evaluation of risk (and so approval pathways) can depend as much on a device’s established safety record as its features.
Tolerability is sometimes distinguished from safety. Something that is bothersome, either transiently or in a minor way, may be a tolerability issue. Any irreversible damage to tissue or permanent impairment in function is a safety issue. A side-effect implies any effects from the device other than the main intended use. Those side-effects that are undesirable, such as nausea, can also be called an adverse event. A serious adverse event is an outcome of device use that results in death, is life threatening, requires hospitalization, or results in disability or permanent damage. If medical intervention is warranted to avoid any of the above, it would also be a serious adverse event. It is possible for a person in a clinical trial to have a (serious) adverse event that is unrelated to the device itself (is incidental) and it is important to understand when events are related to device use or not. A device causing a serious adverse event would be a significant risk and present safety concerns.
The implantation of any device is associated with some risk. Evidently, an opening must be made in the skin and any other tissues (e.g., skull) leading to the implant location. When inserting any object inside the brain, some damage is inevitable. It is not the case as one neurosurgeon mused that “the neurons move out of the way of the implanted electrode”. No matter how small the electrodes or clever the implantation procedure (such as Neuralink’s robot surgeon) a minimal amount of damage (injury) is expected. Indeed, I suggested with colleagues that an implanted electrode may not only injure local tissue, but harm distant connected brain regions.
The FDA can approve a medical device even with risk of serious adverse event provided the overall benefits of using the device outweigh the risks. In fact, all FDA approvals are based on risk vs benefit. And most medical device regulations and standards relate to ways to identify and manage risk.
For non-invasive devices, a possible goal is to be non-significant-risk: No established concerns for safety, tissue damage, or serious adverse events. Transcranial Direct Current Stimulation (tDCS, a form of non-invasive electrical stimulation), for example, is considered safe by this standard, as would almost any wearable brain monitoring device. Implanted devices have the risk of the surgical procedure itself and then any hazardous interactions of the device with the body. Any interactions of non-invasive or invasive devices with external devices (e.g., MRI) can represent a safety risk as well. Some risks are managed by instructions to the patient and care-giver (e.g., do not go in an MRI with the device). Some non-invasive devices (those that used higher energy levels) may be associated with significant risk, but this is typically rare events (such as seizures by Transcranial Magnetic Stimulation, TMS) or acute side-effects (such as short term disorientation by Electroconvulsive Therapy, ECT) – and not associated with issue injury.
Dr. Parag Patil explained what safety meant to him like this:
Is a medical intervention with a 1% (1 in 100) risk of death “safe”? What about 1 in 200,000 risk of death, which is about the risk with general anesthesia? Or a 0.0001% (one in a million) risk of minor but lasting impairment? There is an inescapable relativity in how people think about value laden terms like “safety”. In the end, the human mind has a hard time thinking about low-probability but catastrophic risks, like a plane crashing or dying during surgery. It’s anxiety provoking, and most patients and doctors have a hard time managing it.
Is there an inherent trade-off between safety (or risk) and efficacy? For example, is an invasive device like DBS necessarily more effective than a non-invasive device like tDCS? This kind of thinking is wrong. A technology can theoretically be both highly unsafe and ineffective. Conversely, there are endless examples of both high-risk and low-risk devices providing a patient’s transformative benefit (see Dr. Vince Clark’s talk on ‘Small Medicine’). However, when a device is associated with increasing risk, the bar for efficacy (potential benefit or quality of evidence) should also be raised. This is precisely the risk vs benefit analysis – even when such decisions are subjective or difficult. It is also misguided to assume a therapy that provides benefits must also produce a deficit – under some notion that brain function is ’zero sum.’
In summary, in neurotechnology the term safe should be understood in context. For non-invasive neurotechnology that records brain activity, safe implies low risk or non-significant-risk (no risk of a serious adverse event or tissue damage). For non-invasive technologies that stimulate the brain, low-intensity devices are typically non-significant risk, with minor and acute tolerability risks (e.g., short-term skin redness). For non-invasive technologies that use high intensities, risks may increase (without evidence of lasting injury). For implanted neurotechnology devices, safe implies devices and procedures minimizing tissue damage and the risk of a serious adverse event, which is the goal of minimally-invasive devices. But in all cases, what is safe is relative and contextual, depending on the exact technology and methods of use, patient selection, and level of (developing) evidence.
Limits
In the first part of “What Silicon Valley needs to know about the brain, and what it doesn't”, I considered the ambitions of consumer-directed implanted neurotechnologies. The reach can span all the way to healthy individuals enhancing their brain function, while typically starting with patients with severe paralysis. Patients with so much loss in function they are in danger of becoming ‘locked in.’ This strategy makes sense, given the risk vs benefit calculation at each stage of device development. Safety considerations depend on evidence, and so perception of risk decreases with success in initial patients (which is why clinical trials proceed in expanding phases). But it puzzles me why the need for a device with all the bells-and-whistles for this first use case? Why not make a device that is, for example, twice as big and with a fraction of the electrode number (but still may be more than in existing applications) to establish safety and efficacy in these patients? Or is this over-design part of the Silicon Valley technology model? It would seem companies that have made clinical headway (like Blackrock Neurotech and Sychron) have instead focused on technology for short term - but still transformative - wins.
Indeed, are technologies will less channels (electrodes) or that are non-invasive inherently disadvantaged compared to an implanted device with hundreds of channels? That argument underpins the sales pitch of companies like Neuralink. But I think that's like arguing more wheel and horsepower necessarily make a better car – surely that depends on where you plan to drive? One cannot divorce the neurotechnology use-case with the hardware requirements. In the first part of “What Silicon Valley needs to know about the brain, and what it doesn't”, I explained that ‘smaller/more electrodes’ and ‘better software’ where the technology levers technologists can pull – but how this would necessarily lead to uses was a mystery. From Part 2, we see there are tradeoffs. The image below shows that any technology with many dense electrodes cannot span the entire brain; other technologies can span much of the brain and rely on recording aggregate brain activity; and still other technologies (such as DBS and SCS) have had great success in a middle ground by matching to an appropriate use-case.

Note: Nothing in this essay, or referenced materials, should be taken as technical, medical, or legal advice.
Acknowledgements: I want to thank Nigel Gebodh, Abhishek Datta, and Parag Patil for valuable discussions and editing.
My new essay "What Silicon Valley needs to know about the brain, and what it doesn't. Part 2: Definitions"
— Marom Bikson (@MaromBikson) January 12, 2023
What does 'electrode', 'minimally-invasive', 'non-invasive' & 'safe' mean in neurotechnolgy development for brain recording (BCI) & neuromodulation?https://t.co/roR5Uod3fA
Read this next
What Silicon Valley needs to know about the brain, and what it doesn't - Part I
Silicon Valley wants to get inside your head - literally. While these projects adopt mixes of commercial stealth and elaborate publicity, and the breadth of applications spans the entire human condition, all these technologies take one of two product forms: Either as a chip implanted in your head transmitting to a computer or as a wearable cap transmitting to a computer.
Neuromodulation for COVID-19, Neuromodec update 7: Electronic noses, NIH “RECOVER” recruits’ patients, New prospects.
This is the 7th update from Neuromodec (August 5, 2022) on the use of Neuromodulation for COVID-19 as part of Neuromodec’s initiative to educate on and advance treatments.
NYU Langone Hospital Announces At-Home Virtual Brain Stimulation Program
The NYU Langone Health medical system announced at-home service for non-invasive (wearable) neuromodulation...